You are here
Frequently Asked Questions (FAQ) - SME-Instrument
SME-General (1)
An application for a collaborative project is not interfering with an application for the SME instrument.
Yes, the restriction is only WITHIN the SME instrument
As subcontractor that MIGHT be possible, but as PARTNER definitely NOT
The high growth potential is intended to help Europe achieve the EUROPE 2020 goals which is: jobs, jobs, jobs!
SME-Phase 2 (1)
A complementary funding is not excluded in the work programme.
The 50.000 EUR can be used for any kind of activity (personnel costs, subcontracting, costs for patent search etc).
Yes for the PHC topic, funding rate is 100%
Please use the definitions as shown here (and please see also the corresponding literature)
A biomarker is a characteristic that is objectively measured and evaluated as an indicator of normal biologic processes, pathogenic processes, or pharmacologic responses to a therapeutic intervention (NHI Biomarkers Definitions Working Group; 2001)
A valid biomarker is defined as “a biomarker that is measured in an analytical test system with well-established performance characteristics and for which there is an established scientific framework or body of evidence that elucidates the physiologic, toxicological, pharmacologic, or clinical significance of the test results
(FDA. Guidance for industry - pharmacogenomic data submissions. 2005)
Air Force 1SME-Submission (4)
They will be rejected with the possibility to resubmit an improved version.
Yes. You can download it under http://ec.europa.eu/research/participants/data/ref/h2020/call_ptef/pt/h2020-call-pt-sme-1_en.pdf
Accesorios para el runningAny SME must be (re)checked, if it was not validated within the last two years.
SME-Evaluation (3)
At least 2 evaluators are planned to evaluate a proposal.
SME-Reporting (2)
Yes, reporting periods such as once a year; the general rules H2020 apply.
EASME is working on a database that is not ready yet. EEN will be included in this.
The SME instrument is targeted at all types of innovative SMEs showing a strong ambition to develop, grow and internationalise. It provides staged support covering the whole innovation cycle in three phases complemented by a mentoring and coaching service. Transition from one phase to the next will be seamless provided the SME project proves to be worth further support in a further evaluation. Each phase is open to new entrants.
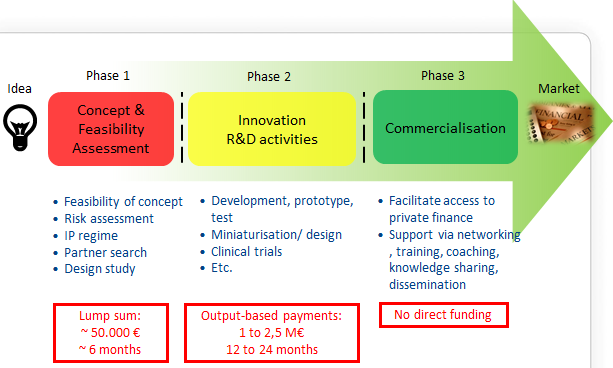
a) SME instrument (phase 1)
Description: Feasibility study verifying the technological/practical as well as economic viability of an innovation idea/concept with considerable novelty to the industry sector in which it is presented (new products, processes, design, services and technologies or new market applications of existing technologies). The activities could, for example, comprise risk assessment, market study, user involvement, Intellectual Property management, innovation strategy development, partner search, feasibility of concept and the like to establish a solid high-potential innovation project aligned to the enterprise strategy and with a European dimension. Bottlenecks in the ability to increase profitability of the enterprise through innovation shall be detected and analysed during phase 1 and addressed during phase 2 to increase the return in investment in innovation activities.
Funding rate: Funding will be provided in the form of a lump sum of EUR 50,000 (70% funding rate).
b) SME instrument (phase 2)
Description: innovation projects that address a specific challenge and demonstrate high potential in terms of company competitiveness and growth underpinned by a strategic business plan. Activities should focus on innovation activities such as demonstration, testing, prototyping, piloting, scaling-up, miniaturisation, design, market replication and the like aiming to bring an innovation idea (product, process, service etc.) to industrial readiness and maturity for market introduction, but may also include some research. SMEs can subcontract or buy in work and knowledge that is essential for their innovation project in the spirit of the innovation voucher concept. Proposals should be based on a strategic business plan either developed through phase 1 or another means.
Funding rate: 70% [or in exceptional cases, where the research component is predominant, the reimbursement rate may be 100%, as specified in each topic].
c) SME instrument (phase 3)
Support to commercialisation promotes the wider implementation of innovative solutions and customers and supports financing of growth by facilitating access to public and private risk capital. This stage will not provide for direct funding, but SMEs can benefit from indirect support measures and services as well as access to the financial facilities supported under Horizon 2020.
d) Mentoring and coaching
Each beneficiary of the SME instrument will be offered business coaching support during Phase 1 (up to 3 coaching days) and Phase 2 (up to 12 coaching days) in addition to the grant offered. This support will be provided through the Enterprise Europe Network (EEN) and delivered by a group of qualified and experienced business coaches. The local EEN office will introduce the beneficiary to the coaching process and propose a selection of coaches from the database managed by the Commission for the beneficiary to choose from. The objective is to accelerate the impact of the support provided through the SME instrument and to equip beneficiaries with the necessary skills, business processes and relevant competencies for long-term growth. Phase 3 does not include individual business coaching, but SME instrument participants will be able to count on continuing EEN support in linking to relevant support services within the Network, regionally or nationally. It is important to note that the objective of coaching is not to support the company in project management or reporting obligations related to Horizon 2020 participation.
ADIDASContact Info
Austrian Research Promotion Agency
- Sensengasse 1, A-1090 Vienna, AUSTRIA
- infofitforhealth.eu

